Translate this page into:
Unveiling Leprosy Trends through the COVID-19 Lens: A 6-Year Analysis
*Corresponding author: Rahul Suresh Nayak, Department of Dermatology, Venereology and Leprosy, Hassan Institute of Medical Sciences, Hassan, Karnataka, India. drrahulnayak19@gmail.com
-
Received: ,
Accepted: ,
How to cite this article: Nayak RS, Rangaswamy UH, Rangegowda SM, Nagesha PC, Channakeshavaiah RB, Nirvanappa VK, et al. Unveiling Leprosy Trends through the COVID-19 Lens: A 6-Year Analysis. Indian J Postgrad Dermatol. doi: 10.25259/IJPGD_241_2024
Abstract
Objectives
This study analysed clinical and epidemiological patterns of leprosy over 6 years, focusing on three periods: before (2018–2019), during (2020–2021) and after COVID 19 pandemic (C19P) (2022–2023).
Materials and Methods
Retrospective data, including demographic, clinical and treatment details, were collected from the district leprosy officer.
Results
Of 92 cases, 67 (72.8%) were male and 25 (27.2%) female, with most (54, 58.6%) aged 15–45, and 5 (5.4%) childhood cases. Local residents accounted for 78 cases (84.7%) and migrants 14 (15.3%). Multibacillary leprosy was prevalent (76, 82.6%), with borderline lepromatous as the most common type (37, 39.6%). Lepra reactions (LRs) occurred in 13 cases (8 T1R, 5 T2R), and 31 (34.6%) had deformities, mainly Grade 1 disabilities in 21 cases (22.8%). During COVID-19 (2020–2021), cases dropped from 44 to 12, with a relative increase in deformities. Post-C19P (2022–2023) showed 35 cases, 9 LRs and 17 deformities, marking a 20% rise in reactions and deformities from pre-C19P.
Conclusion
C19P disrupted leprosy management, leading to more deformities and LRs. Attention to coinfections is crucial, as viral infections can worsen leprosy outcomes, underscoring the need for vigilant care during outbreaks.
Keywords
Coinfections
COVID-19 pandemic
Deformities
Hansen’s disease
Lepra reactions
Leprosy
INTRODUCTION
Leprosy is a chronic infectious and granulomatous disease resulting from Mycobacterium leprae. The disease primarily impacts the skin, peripheral nerves, mucosal surfaces of the upper respiratory tract and the eyes.[1] Leprosy, eradicated globally in 2000 and eliminated from India in 2005 (prevalence <1/10,000), remains a public health issue in some endemic regions. In 2019, 202,256 new cases were reported worldwide, including 14,893 among children and 10,816 with Grade 2 Disability (G2D).[2] India currently maintains a prevalence rate of 0.41, with 5.76% of cases affecting children and 2.41% of them resulting in G2D.[3] Although leprosy was no longer a public health concern in Karnataka after 2005, new cases persist, with a prevalence rate of 0.3/10,000 population.[4]
The COVID-19 pandemic (C19P) (2020–2021) disrupted global healthcare, delaying diagnoses of neglected diseases like leprosy. In 2020, global leprosy data showed a significant decline in total registered cases (TRC) to 129,152, including a 37.1% drop in new cases, a 2% decrease in G2D cases and a 0.6% reduction in childhood cases.[2] COVID-19 and M. leprae coinfection increased interleukin (IL)-6 and IL-12 levels, potentially intensifying inflammation.[5] A study in Himachal Pradesh found a post-COVID-19 increase in new leprosy cases, leprosy reactions (LRs) and disabilities.[6] A population-level study on COVID-19’s impact on leprosy reported a significant decrease in diagnoses but a rise in multibacillary (MB) cases.[7] This study aimed to examine the clinicoepidemiological aspects of leprosy and assess the impact of the C19P on leprosy in a district of Southern Karnataka.
MATERIALS AND METHODS
A retrospective cross-sectional study at a tertiary centre in Southern Karnataka analysed data from January 2018 to December 2024, with the Institutional Ethics Committee approval. Diagnosis was based on any of the three cardinal signs of leprosy.[8]
The study included newly diagnosed leprosy cases and patients who defaulted or relapsed and were restarted on multi-drug therapy (MDT). Patients with unrelated deformities or incomplete records were excluded from the study. Demographics, physical findings and clinical diagnoses were documented. Leprosy was classified per the RidleyJopling system: Tuberculoid (TT), Borderline Tuberculoid (BT), Borderline (BB), Borderline Lepromatous (BL) and Lepromatous (LL).[9] The deformities were classified into Grades 0, 1 and 2 according to the WHO disability grading system.[10] For treatment purposes, these cases were classified into PB and MB types according to the WHO criteria.[11]
Statistical analysis
Data were entered into Microsoft Excel and analysed using the Statistical Package for the Social Sciences software, categorised into three periods: Pre-COVID-19 (PreC19P), COVID-19 (C19P) and post-COVID-19 (PostC19P). Results were presented as frequencies and percentages.
RESULTS
During the study, 92 TRC were recorded, including 67 males and 25 females. Childhood leprosy constituted 5.4% of cases. Most patients were aged 15–45 years (mean age: 42.1 years). Local inhabitants made up 84.7% (78 cases), primarily from Southern Karnataka districts, with Hassan reporting the highest cases (72 overall, 26 from Hassan Taluk). Migrants from Bihar, Jharkhand, West Bengal and Andhra Pradesh accounted for 15.3% (14 cases) [Table 1 and Figure 1].
| Characteristics | n (%) |
|---|---|
| Gender | |
| Males | 67 (72.8) |
| Females | 25 (27.2) |
| Children (age group in years) | |
| 0–18 | 5 (5.4) |
| 19–30 | 23 (25) |
| 31–45 | 29 (31.5) |
| 46–60 | 20 (21.7) |
| >60 | 15 (16.3) |
| Residents | |
| Natives | 78 (84.7) |
| Migrants | 14 (15.3) |
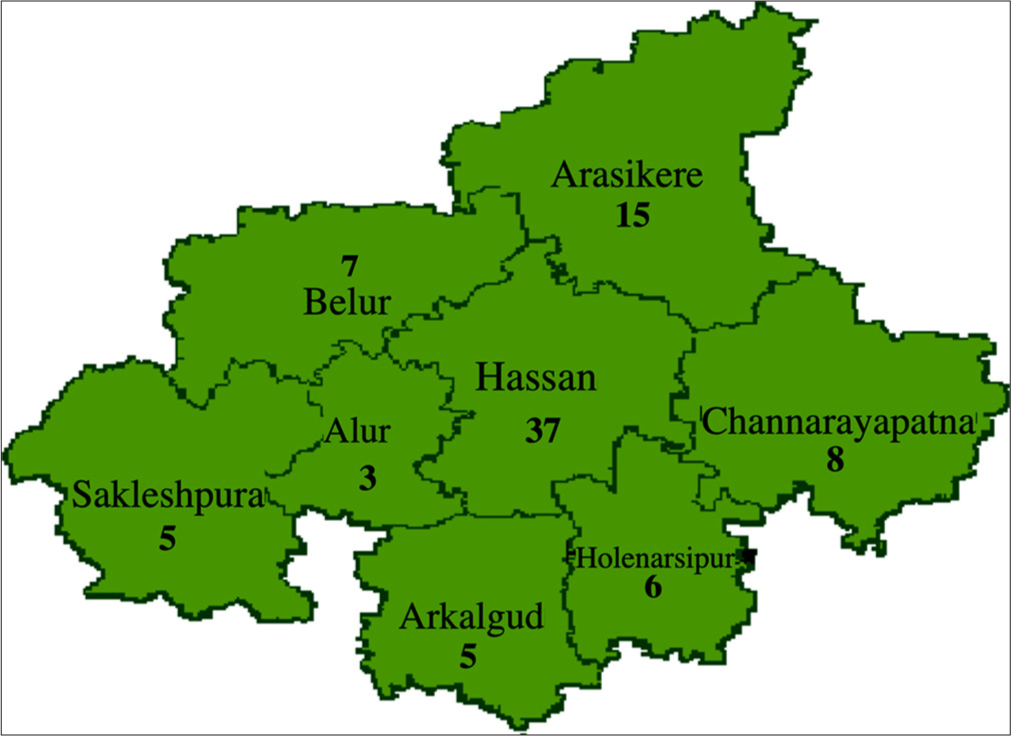
- Distribution in Hassan District.
According to the disease spectrum, the majority of cases were BL, followed by BT and LL. The proportion of MB cases was higher than that of PB cases [Table 2].
| PB/MB | n (%) | Leprosy spectrum | n (%) |
|---|---|---|---|
| PB | 16 (17.3) | TT | 5 (5.4) |
| BT | 29 (31.5) | ||
| MB | 76 (82.6) | BL | 37 (40.2) |
| LL | 16 (17.3) | ||
| Pure neuritic | 3 (3.2) | ||
| Histoid Hansen | 2 (2.1) |
TT: Tuberculoid, BT: Borderline tuberculoid, BL: Borderline lepromatous, LL: Lepromatous, PB: Paucibacillary, MB: Multibacillary.
LRs were observed in 13 cases (14.3%), with 8 cases (8.8%) being Type 1 Reaction (T1R) and the rest Type 2 Reaction (T2R). T1R was primarily noted in the TT spectrum (four cases, 4.3%), while T2R occurred in the BL (three cases, 3.3%) and LL (two cases, 2.1%) spectra.
Deformities were identified in 31 cases (34.6%), with 21 (22.8%) having Grade 1 Disability (G1D). Among local inhabitants, 22 cases (28.2%) had disabilities, including 13 with G2D. Immigrants had 4 cases (28.5%) with disabilities, mostly G1D (3 cases). While deformities were proportionally higher in immigrants, G2D was more prevalent among locals. In addition, 3 relapse cases (3.2%) were recorded.
Effects of the C19P
The C19P significantly impacted leprosy trends, as reflected in the TRC. Pre-COVID-19 (2018–2019), 44 cases were recorded, while 13 cases were registered during the pandemic (2020–2021), and 19 and 17 cases were reported in 2022 and 2023, respectively, in the post-pandemic period.
The predominant leprosy spectrum shifted across the periods: BL (23 cases, 52.2%) pre-pandemic, LL (6 cases, 50%) during the pandemic and BT (13 cases, 36.1%) post-pandemic. No significant differences were noted in MB and paucibacillary (PB) cases across the periods. Lepra reactions (LRs) and disabilities showed a rising trend post-pandemic. LRs increased from 2 cases (4.5%) pre-pandemic to 2 cases (16.6%) during the pandemic and 9 cases (25%) post-pandemic. Disabilities rose from 12 cases (27.2%) pre-pandemic and 3 cases (25%) during the pandemic to 17 cases (47.2%) post-pandemic, the highest recorded. Post-pandemic increases in LRs and deformities were 20.5% and 20%, respectively, compared to pre-pandemic levels [Table 3].
| Time periods | No reactions - n (%) | Type 1 n (%) | Type 2 n (%) | P-value | |
| Lepra reactions | Pre-COVID | 42 (53.2) | 1 (12.5) | 1 (20.0) | 0.043 |
| COVID | 10 (12.6) | 2 (25.0) | 0 (0.0) | ||
| Post-COVID | 27 (34.2) | 5 (62.5) | 4 (80.0) | ||
| Time periods | No deformities - n (%) | Grade 1 n (%) | Grade 2 n (%) | P-value | |
| Deformities | Pre-COVID | 32 (53.3) | 6 (28.6) | 6 (54.5) | 0.251 |
| COVID | 9 (15.0) | 2 (9.5) | 1 (9.1) | ||
| Post-COVID | 19 (31.7) | 13 (61.9) | 4 (36.4) |
No means absence of lepra reaction or absence of deformity
The proportion of cases with LRs was highest in the post-COVID-19 period (Post-C19P), with 9 cases (25% of all cases). T1Rs were slightly more common than T2R. The difference in the incidence of LRs across the three periods was statistically significant (P = 0.043) [Figure 2].
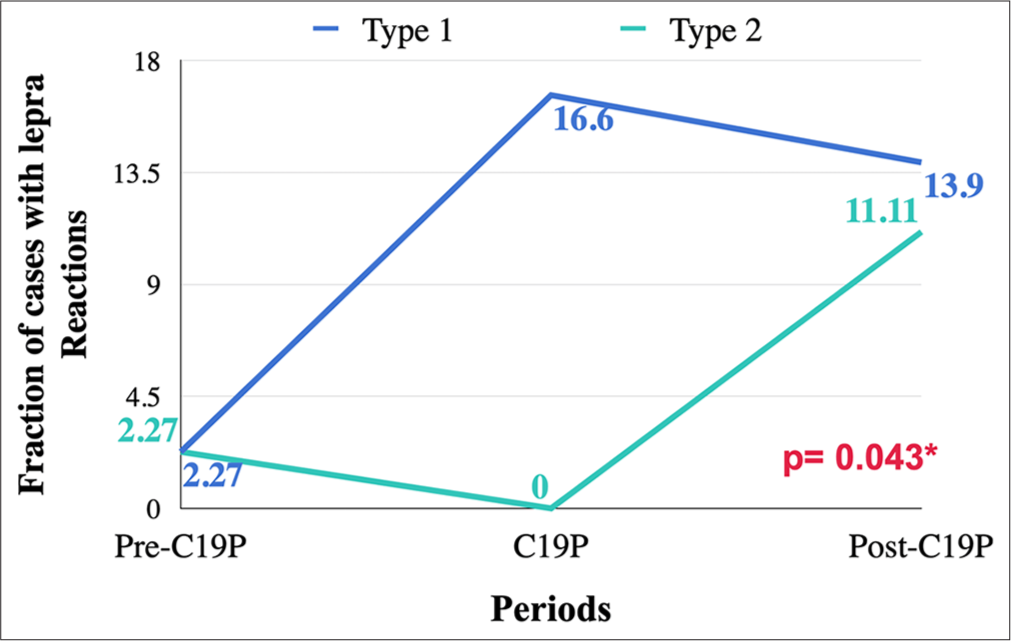
- Lepra reactions % (out of total cases in each respective period). *Statistically significant.
The proportion of cases with deformities was also highest in post-C19P, with 17 cases (47.2% of all cases). Most deformities were Grade 1. However, the difference in the incidence of deformities across the three periods was not statistically significant (P = 0.251) [Figure 3].
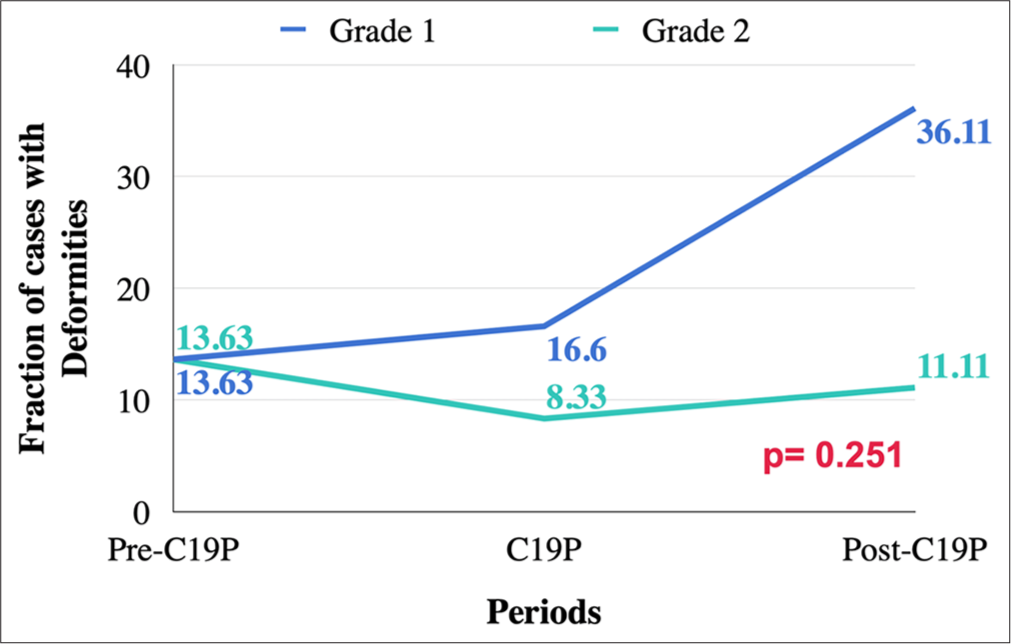
- Deformities % (out of total cases in each respective period).
DISCUSSION
Leprosy, once considered incurable at the 1st International Congress in Berlin, saw a shift with the identification of M. leprae as its causative agent in 1873. This discovery, along with the therapeutic benefits of chaulmoogra oil and later dapsone, sparked hope for effective treatment and management of the disease.[12] MDT became widely used in 1982, and the National Leprosy Eradication Programme was launched in 1983. As a result, leprosy was eliminated from India in 2005.[3] Leprosy affects all ages and genders but are more common in males and individuals aged 20–40, likely due to factors such as increased outdoor exposure, greater disease awareness and health-seeking behaviour. This finding is consistent with other studies.[13,14] Most cases were local residents of Hassan and nearby districts, due to the proximity of our centre. The immigrant demographic primarily consisted of individuals from highly endemic regions. Analogous patterns have been observed within our state and in different parts of the country.[15-18]
Childhood leprosy poses serious risks of transmission in school settings. The global childhood leprosy rate in 2020 was 6.2%, with a national rate of 5.77%.[3] However, in our study, this rate was 5.4%.
In our study, 76 (82.6%) of cases presented with MB leprosy. Similar findings of higher MB cases were reported in other studies.[14,19] The high prevalence of MB cases significantly contributes to transmission and increases the risk of reactions and deformities. This also suggests delayed diagnosis, often due to limited healthcare access, particularly during the C19P.
The most frequent morphologic type in our study was the BL 37 (39.6%) followed by the BT 29 (31.9%) spectrum, consistent with a few more studies where most cases were reported in the BT spectrum, then in BL and LL.[19-21] In addition, special types of leprosy were recorded in 5 (5.4%) of patients, including Pure Neuritic Leprosy (PNL) 3 (3.2%) and Histoid 2 (2.1%).
In our study, LRs were observed in 13 (14.3%) cases, lower than that reported by many earlier studies. We observed a higher incidence of T1R with 8 (61.5%) cases when compared to T2R.[6,13,15,22,23]
Our study shows that the impact of the C19P was most evident in the TRC. During the pre-C19P period (2018–2019), normalcy facilitated effective leprosy screening and detection. Functional healthcare systems, public health initiatives and widespread screening in endemic areas enabled prompt case identification and treatment. Regular community interactions with healthcare providers raised awareness, ensuring robust surveillance, consistent funding and accessible resources for efficient leprosy management. These were disrupted severely by the C19P. Thus, the TRC was reduced to a mere 12 cases during the C19P (2020–2021), a 72.7% decline. Public health measures such as lockdowns, social distancing and travel restrictions, while necessary to control the virus, further disrupted daily life and healthcare services. This disruption meant that non-COVID-related health issues, such as leprosy, received less attention and regular health screenings and treatments, were postponed or cancelled.
During the post-C19P period (2022–2023), the TRC increased to 36, likely due to delayed detection during the pandemic. However, this rise remained below pre-pandemic levels, possibly due to ongoing fear of COVID-19, concerns about its long-term effects and hesitancy among patients and healthcare workers to resume routine medical activities.
These findings agree with a population-focused research by Matos et al. which noted a 44.4% decline in leprosy diagnoses when comparing 2019 to 2020 data.[7] However, this is contrary to the findings of Verma et al.[6] where there was a sharp decline in the TRC during the peak followed by a sharp increase in the TRC post-C19P.
The decline in leprosy case detection and delayed treatment during the pandemic likely contributed to the post-COVID-19 surge in LRs and deformities. BB leprosy, which requires timely treatment to prevent reactions and nerve damage, was particularly affected, leading to the highest incidence of LRs and G1D post-pandemic.
Santos Morais Junior et al. found that coinfection with COVID-19 and M. leprae led to elevated levels of IL-6 and IL-12, triggering a stronger inflammatory response. In leprosy, T1R patients show heightened IL-17 release, while T2R patients exhibit strong antibody responses but weaker effector T-cell responses.[5]
Furthermore, COVID-19 vaccines trigger an inflammatory cascade through neutrophilia and heightened Th1 T-cell responses, characterised by increased production of tumour necrosis factor-α and interferon-γ by CD4+ T cells. This elevated cell-mediated immunity against leprosy bacilli may explain the increased incidence of both types of LRs.[24]
These findings suggest a possible link between COVID-19 infection/vaccination and the increased incidence of LRs and G1D post-pandemic. This aligns with Verma et al.,[6] which also reported an increase in deformities and reactions post-COVID-19. COVID-19 may act as a comorbidity, heightening the likelihood of complications in leprosy patients, particularly in LRs.[25]
During the C19P, global leprosy reporting declined, with only 127 countries submitting data in 2020, compared to 160 in 2019, underscoring the impact on control efforts. The study’s limitations include its retrospective design, small sample size and lack of data on COVID-19 coinfection and vaccination. Addressing coinfections is crucial, as viral infections can complicate leprosy’s clinical presentation by triggering immune dysregulation and unmasking subclinical infections due to immunosuppression.
Prevalent knowledge
Leprosy, caused by M. leprae, commonly affects males and individuals aged 20–40, often due to outdoor exposure and health-seeking behaviour
MB cases dominate leprosy diagnoses, with delayed detection increasing the risk of transmission and deformities
The C19P disrupted healthcare services, significantly impacting leprosy detection and management, as seen in global and national trends.
New knowledge
The study highlights a decline in leprosy case detection during the C19P, followed by a surge in post-pandemic cases due to delayed diagnosis
It found potential links between COVID-19 infection/vaccination and increased incidence of LRs and deformities, likely due to enhanced immune responses
The rise in deformities and reactions post-pandemic emphasises the need for timely intervention and monitoring of BB leprosy to prevent severe outcomes.
CONCLUSION
COVID-19 coinfection may exacerbate complications in leprosy patients, increasing LRs. The pandemic caused a sharp decline in registered cases due to reduced detection during lockdowns, followed by a rise in LRs and deformities, likely linked to COVID-19 and healthcare disruptions. Despite this, case numbers remained below pre-pandemic levels post-COVID, reflecting ongoing fear and delays in healthcare access. Given leprosy’s long incubation and treatment periods, enhanced care is essential, as coinfections, particularly viral ones, can trigger immune responses that complicate outcomes.
Acknowledgement
The authors would like to sincerely acknowledge the Institutional Ethics Committee and the Medical Research Unit of the Institute for Plagiarism support.
Ethical approval
The research/study approved by the Institutional Review Board at Hassan Institute of Medical Sciences, number IEC/HIMS/RR549/29-02-2024, dated 29th February, 2024.
Declaration of patient consent
Only statistical patient data is included in the study, due to its retrospective nature, the necessary permission from the District leprosy officer to use the statistical data has been obtained. All patient data has been identity-masked.
Conflicts of interest
There are no conflicts of interest.
Use of artificial intelligence (AI)-assisted technology for manuscript preparation
The authors confirm that there was no use of artificial intelligence (AI)-assisted technology for assisting in the writing or editing of the manuscript and no images were manipulated using AI.
Financial support and sponsorship: Nil.
References
- Global Leprosy (Hansen Disease) Update 2020. Impact of COVID-19 on the Global Leprosy Control. World Health Organization. No 36 2020 Available from: https://www.who.int/publications/i/item/who-wer9636-421-444 [Last accessed on 2024 Jul 15]
- [Google Scholar]
- Annual Report for the Year 2020-2021. 2021. Central Leprosy Division. Directorate General of Health Services, Ministry of Health and Family Welfare, Government of India. Available from: https://dghs.gov.in/content/1349_3_nationalleprosyeradicationprogramme.aspx [Last accessed 2024 Jul 15]
- [Google Scholar]
- Update on Leprosy Cases in the Country. 2023. Available from: https://pib.gov.in/pressreleasepage.aspx?prid=1909081 [Last accessed on 2024 Jul 15]
- [Google Scholar]
- Circulating Interleukin-6 (but not Other Immune Mediators) Associates with Criteria for Fried's Frailty Among Very Old Adults. J Aging Res. 2020;2020:6831791.
- [CrossRef] [PubMed] [Google Scholar]
- Clinico-Epidemiological Trends of Leprosy in 21st Century and During COVID-19 Pandemic. Indian J Leprosy. 2022;94:299-308.
- [Google Scholar]
- Impact of the COVID-19 Pandemic on the Diagnosis of New Leprosy Cases in Northeastern Brazil, 2020. Int J Dermatol. 2021;60:1003-6.
- [CrossRef] [PubMed] [Google Scholar]
- Leprosy. An Update: DEFINITION, Pathogenesis, Classification, Diagnosis, and Treatment. Actas Dermosifiliogr. 2013;104:554-63.
- [CrossRef] [PubMed] [Google Scholar]
- Classification of Leprosy According to Immunity: A Five-Group System. Int J Lepr Other Mycobact Dis. 1966;34:255-73.
- [Google Scholar]
- WHO Disability Grading: Operational Definitions. Lepr Rev. 2003;74:366-73.
- [CrossRef] [Google Scholar]
- Pauci-and Multibacillary Leprosy: Two Distinct, Genetically Neglected Diseases. PLoS Negl Trop Dis. 2016;10:e0004345.
- [CrossRef] [PubMed] [Google Scholar]
- Clinico-epidemiological Profile of Leprosy in Post Elimination Era: A Hospital-based Study. Indian J Lepr. 2019;91:197-205.
- [Google Scholar]
- Leprosy in the Post-elimination Era: A Clinico-epidemiological Study from a Northern Indian Tertiary Care Hospital. Acta Dermatovenerol. 2019;28:7-10.
- [CrossRef] [Google Scholar]
- Trends in Profile of Leprosy Cases Reporting to a Tertiary Care Centre in Delhi During 2006-2015. Indian J Lepr. 2016;88:217-25.
- [Google Scholar]
- Current Scenario and Challenges of Urban Leprosy in a Tertiary Care Regional Centre in Western India: A 5-Year Observational Retrospective Study. Indian J Lepr. 2017;89:1-7.
- [Google Scholar]
- A Clinico-Epidemiological Scenario of Leprosy at a Tertiary Care Centre in Sub-Himalayan Region: A Seven-year Retrospective Study. Indian J Lepr. 2019;91:7-16.
- [Google Scholar]
- Clinical and Epidemiological Characteristics of Leprosy Patients in the post Elimination Era: We Need to be Vigilant. Indian J Lepr. 2021;93:63-75.
- [Google Scholar]
- Epidemiological Trends of Leprosy in An Urban Leprosy Centre of Delhi: A Retrospective Study of 16 Years. Indian J Lepr. 2011;83:201-8.
- [Google Scholar]
- A retrospective clinico-epidemiological study of leprosy cases treated at a tertiary care hospital in Western Maharashtra. Med J Dr. D.Y. Patil Vidyapeeth. 2021;14:385-91.
- [CrossRef] [Google Scholar]
- Trends in Detection of New Leprosy Cases at Two Centres in Himachal Pradesh, India: A Ten-year Study. Indian J Lepr. 2003;75:17-24.
- [Google Scholar]
- Leprosy scenario at a tertiary level hospital in Delhi: A 5-year retrospective study. Indian J Dermatol. 2015;60:55-9.
- [CrossRef] [PubMed] [Google Scholar]
- Five-Year Experience with Type 1 and Type 2 Reactions in Hansen Disease at a US Travel Clinic. Am J Trop Med Hyg. 2008;79:452-4.
- [CrossRef] [PubMed] [Google Scholar]
- COVID-19 Vaccine BNT162b1 Elicits Human Antibody and TH1 T Cell Responses. Nature. 2020;586:594-9.
- [CrossRef] [PubMed] [Google Scholar]
- Epidemiological Study of Co-infection of Leprosy and COVID-19 in the state of Mato Grosso, 2020. Indian J Lepr. 2020;96:57-68.
- [Google Scholar]







