Translate this page into:
Clinical and Histopathological Insights into Adult-Onset Unilateral Nevus of Ota
*Corresponding author: Anshuman Dash, Department of Dermatology and Sexually Transmitted Diseases, Vardhman Mahavir Medical College and Safdarjung Hospital, New Delhi, India. anshu.sipun@gmail.com
-
Received: ,
Accepted: ,
How to cite this article: Dash A, Kathuria S, Sharma S. Clinical and Histopathological Insights into Adult-Onset Unilateral Nevus of Ota. Indian J Postgrad Dermatol. doi: 10.25259/IJPGD_235_2024
Abstract
Dermal melanocytoses are characterised by the presence of spindle-shaped melanocytes in the dermis. The common forms, such as Mongolian spots, and nevi of Ota or Ito, present from birth or puberty, while acquired dermal melanocytoses appear in adulthood and are rare. They are frequently misdiagnosed as lichen planus pigmentosus or melasma leading to delay in treatment. This study reports five cases of acquired unilateral nevus of Ota from India, with an analysis of their dermoscopic and histopathological features.
Keywords
Acquired bilateral nevus of Ota-like macules
Dermal melanocytosis
Dermoscopy
Hori’s nevus
Nevus of Ota
INTRODUCTION
Dermal melanocytoses are a group of diverse entities with the common theme of an intradermal population of dendritic spindle shaped melanocytes. The presentation can be since birth or during puberty for Mongolian spot, Nevus of Ota (NOO) or Nevus of Ito. While Mongolian spot disappears soon after birth, NOO and Ito tend to persist. A second categorisation is the Acquired Dermal Melanocytosis (ADM) which presents during adulthood and is extremely rare. These include Hori’s nevus or acquired bilateral nevus of ota like macules (ABNOM) and acquired NOO or the Sun’s nevus. We present a series of five patients with histopathologically proven acquired unilateral NOO, along with their dermoscopic characteristics.
CASE SERIES
Four Indian females and one male with a mean age of 42 years presented with unilateral distribution of bluish-brown discrete to coalescing macules on an otherwise unremarkable face [Figure 1a-f]. The mean duration of the condition was 6 years. There was no history of appearance of lesions during pregnancy, drug intake, preceding trauma or topical application of cosmetic agents before onset of lesions. Other than bluish scleral pigmentation on the same side [Figure 1d] in all five patients, rest ophthalmological examination was normal. Dermoscopic examination was performed using a polarised handheld dermoscope with ×10 magnification (DermLite DL5, San Juan, Capistrano, CA, USA) [Figure 2a]. Histopathological examination was done in all cases and consistently showed presence of dermal melanocytes with long dendritic processes and cytoplasmic melanin pigment between collagen present in the mid and/or deep dermis [Figure 2b and c]. The clinical, dermoscopic and histopathological characteristics of all the cases are presented in Table 1. The patients were diagnosed as having acquired NOO and were subsequently prescribed Q-switched neodymium-doped yttrium–aluminium–garnet (1064 nanometres) laser treatment.
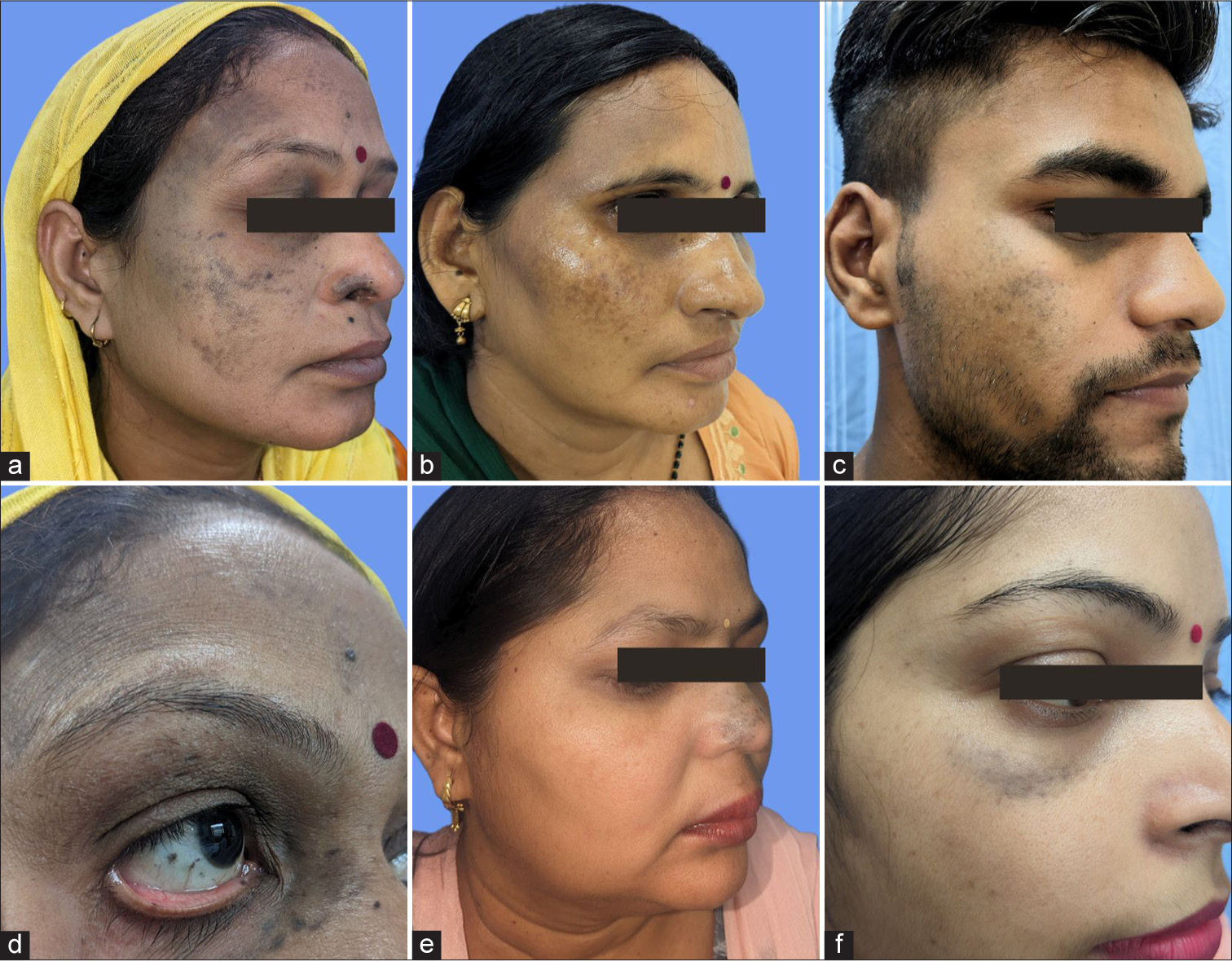
- (a-f) Clinical photos of all cases with unilateral bluish-brown discrete to coalescing macules over the face. (d) Scleral pigmentation is shown.
![(a) Dermoscopic examination showing brown structureless areas with multiple scattered brown dots (black circles) (DermLite DL5, polarised, contact, ×10). (b) Photomicrograph shows unremarkable epidermis. Underlying dermis shows prominent solar elastosis and moderate perivascular and perifollicular chronic inflammation. (haematoxylin and eosin [H&E]; ×100). (c) Photomicrograph shows a high-power view of the elongated dendritic melanocyte (black arrows) with evenly dispersed melanin granules scattered within collagen bundles in mid-dermis. (H&E; ×400).](/content/146/2025/0/1/img/IJPGD-235-2024-g002.png)
- (a) Dermoscopic examination showing brown structureless areas with multiple scattered brown dots (black circles) (DermLite DL5, polarised, contact, ×10). (b) Photomicrograph shows unremarkable epidermis. Underlying dermis shows prominent solar elastosis and moderate perivascular and perifollicular chronic inflammation. (haematoxylin and eosin [H&E]; ×100). (c) Photomicrograph shows a high-power view of the elongated dendritic melanocyte (black arrows) with evenly dispersed melanin granules scattered within collagen bundles in mid-dermis. (H&E; ×400).
| Case | Sex | Age | Clinical findings | Scleral Pigmentation | Dermoscopy | Histopathology |
|---|---|---|---|---|---|---|
| 1 | Female | 45 years | Blue grey macules present on right ala of nose extending to nasal bridge. | Present | Blue-grey structureless areas | Melanocytes in a horizontal distribution in mid-dermis. |
| 2 | Female | 45 years | Blue-grey macules present on right forehead, temple, zygomatic area, cheeks and ala of nose | Present | Blue-grey structureless areas with scattered brown dots | Melanocytes between collagen bundles extending up to mid-dermis |
| 3 | Female | 46 years | Blue-grey macules present on right malar prominence | Present | Brown-grey structureless areas | Melanocytes present between collagen bundles in mid-dermis |
| 4 | Female | 45 years | Brown discrete to coalescing macules on cheeks, zygomatic area and forehead | Present | Brown structureless areas with scattered brown dots | Melanocytes in the mid-dermis |
| 5 | Male | 33 years | Brown to blue discrete to coalescing macules on right cheek and sub-palpebral area | Present | Blue grey structureless areas with scattered brown dots | Melanocytes present between the collagen bundles in the mid dermis. |
DISCUSSION
Melanocytes are descendants of neural-crest cell derivatives called melanoblasts which originate just after closure of neural tube.[1] The melanocytes subsequently start migrating as early as 2.5 weeks in utero and can be demonstrated in the epidermis by 8 weeks.[1] Failure of melanocytes to reach the epidermis or abnormal migration of melanocytes from stratum basale (dropping-off) or from follicular bulbs leads to trapping of melanocytes in the dermis giving rise to dermal melanocytosis.[2] A vast majority of cases of dermal melanocytoses appear in childhood or puberty and are usually diagnosed clinically. However, there exists a rare group of ADM which is clearly adult-onset and frequently misdiagnosed as lichen planus pigmentosus or melasma. The cutoff age for ADM is taken arbitrarily as 25 years based on existing literature. The first studied and well-characterised ADM is Hori’s nevus, described by Hori et al. in 1984 as blue-brown macules involving forehead, cheeks, temples and eyelids bilaterally of Asian women in the third-fourth decade. It typically spares the eye and oral mucosae.[3] The next entity under this classification is the acquired unilateral NOO, of which only 14 cases have been reported in the literature previously. Here, the bluish-grey macules present along the distribution of ophthalmic (V1) or maxillary (V2) distribution of the trigeminal nerve unilaterally. The scleral mucosa is frequently involved. The pathogenesis behind ADMs has been poorly studied. One possible mechanism could be the presence of latent dermal melanocytes which are present unnoticed since birth. Later on, melanin synthesising pathway is activated by ultraviolet radiation, local inflammation, sex hormones or unknown ageing stimuli resulting in pigmentation.[4] Another mechanism could be the presence of extracellular sheath around dermal melanocytes which confer it protection and stability.[4] This sheath has been found to be less developed in Mongolian spots resulting in the regression of pigmentation whereas in NOO, the sheath is thick causing pigmentation to persist.[4]
There exists no difference in the distribution of spindle-shaped melanocytes in the dermis since both acquired unilateral NOO and Hori’s nevus show melanocytes in upper and middermis. Likewise in our cases, we found that the dermal melanocytes are present in the mid-dermis in the majority of the cases which correlates with the blue and grey structureless areas on dermoscopy. This is in contrast to a previous study which found that the melanocytes are present throughout the dermis extending to the dermal-subcutaneous junction.[5] Hence, the distribution of melanocytes in the dermis cannot be reliably used to differentiate between Hori’s nevus and late-onset unilateral NOO. Table 2 provides a brief summary of existing reports on late-onset unilateral NOO.[6-12]
| Year | Authors | Case (s) | Age of onset (average, years) | Ethnicity | Site | Mucosal involvement | Histopathology |
|---|---|---|---|---|---|---|---|
| 1991 | Whitemore et al.[6] | 1 | 42 | Caucasian | Left temple and cheek | Absent | Not available |
| 1994 | Lynn et al.[7] | 1 | 79 | Caucasian | Left cheek, forehead and scalp | Absent | Not available |
| 1999 | Lee et al.[8] | 1 | 70 | Korean | Left periorbital, temple, forehead and scalp | Absent | Melanocytes in upper reticular dermis. |
| 2002 | Chang et al.[9] | 2 | 40 | Korean | Periorbital area, forehead, nose and cheeks | Absent | Melanocytes in upper to mid-dermis |
| 2013 | Quenan et al.[10] | 1 | 32 | Iraqi | Left eyelid, cheek and forehead | Present | Melanocytes in papillary dermis |
| 2019 | Khurana et al.[5] | 6 | 38 | Indian | Unilateral blue-grey macules along V1/V2 distribution | Present in three cases | Melanocytes throughout the dermis extending up to the dermal subcutaneous junction |
| 2019 | Irving Llibran et al.[11] | 1 | 33 | Hispanic | Periorbital and right temporal | Absent | Melanocytes in reticular dermis |
| 2023 | Gupta et al.[12] | 1 | 34 | Indian | Right forehead, cheek and nose | Present | Not performed |
NOO: Nevus of Ota
CONCLUSION
The psychological impact of NOO on patients is immense, which warrants prompt treatment. Due to its rarity in adults, it is crucial for dermatologists to be aware of this condition.
Ethical approval
Institutional Review Board approval is not required.
Declaration of patient consent
The authors certify that they have obtained all appropriate patient consent.
Conflicts of interest
There are no conflicts of interest.
Use of artificial intelligence (AI)-assisted technology for manuscript preparation
The authors confirm that there was no use of artificial intelligence (AI)-assisted technology for assisting in the writing or editing of the manuscript and no images were manipulated using AI.
Financial support and sponsorship: Nil.
References
- The Melanocyte. Its Structure, Function, and Subpopulations in Skin, Eyes, and Hair. Dermatol Clin. 1988;6:161-73.
- [CrossRef] [PubMed] [Google Scholar]
- Late-onset Ito's Nevus: An Uncommon Acquired Dermal Melanocytosis. J Cutan Pathol. 2007;34:640-3.
- [CrossRef] [PubMed] [Google Scholar]
- Acquired, Bilateral Nevus of Ota-like Macules. J Am Acad Dermatol. 1984;10:961-4.
- [CrossRef] [PubMed] [Google Scholar]
- Dermal Melanocytosis: A Clinical Spectrum. Australas J Dermatol. 1996;37:19-25.
- [CrossRef] [PubMed] [Google Scholar]
- Late-onset Naevus of Ota: A Case Series of Six Patients. Clin Exp Dermatol. 2019;44:703-5.
- [CrossRef] [PubMed] [Google Scholar]
- Two Cases of Late Onset Ota's Naevus. Clin Exp Dermatol. 2002;27:202-4.
- [CrossRef] [PubMed] [Google Scholar]
- Late Onset Nevus of Ota: A Rare Presentation. J Am Acad Dermatol. 2019;81:AB231.
- [CrossRef] [Google Scholar]
- Adult Onset Nevus of Ota: Dermoscopic Characterization of A Rare Entity-A Case Report. Apollo Med. 2023;20:269-71.
- [CrossRef] [Google Scholar]







